Now enrolling
See if the PiNACLE trial is
right for you.
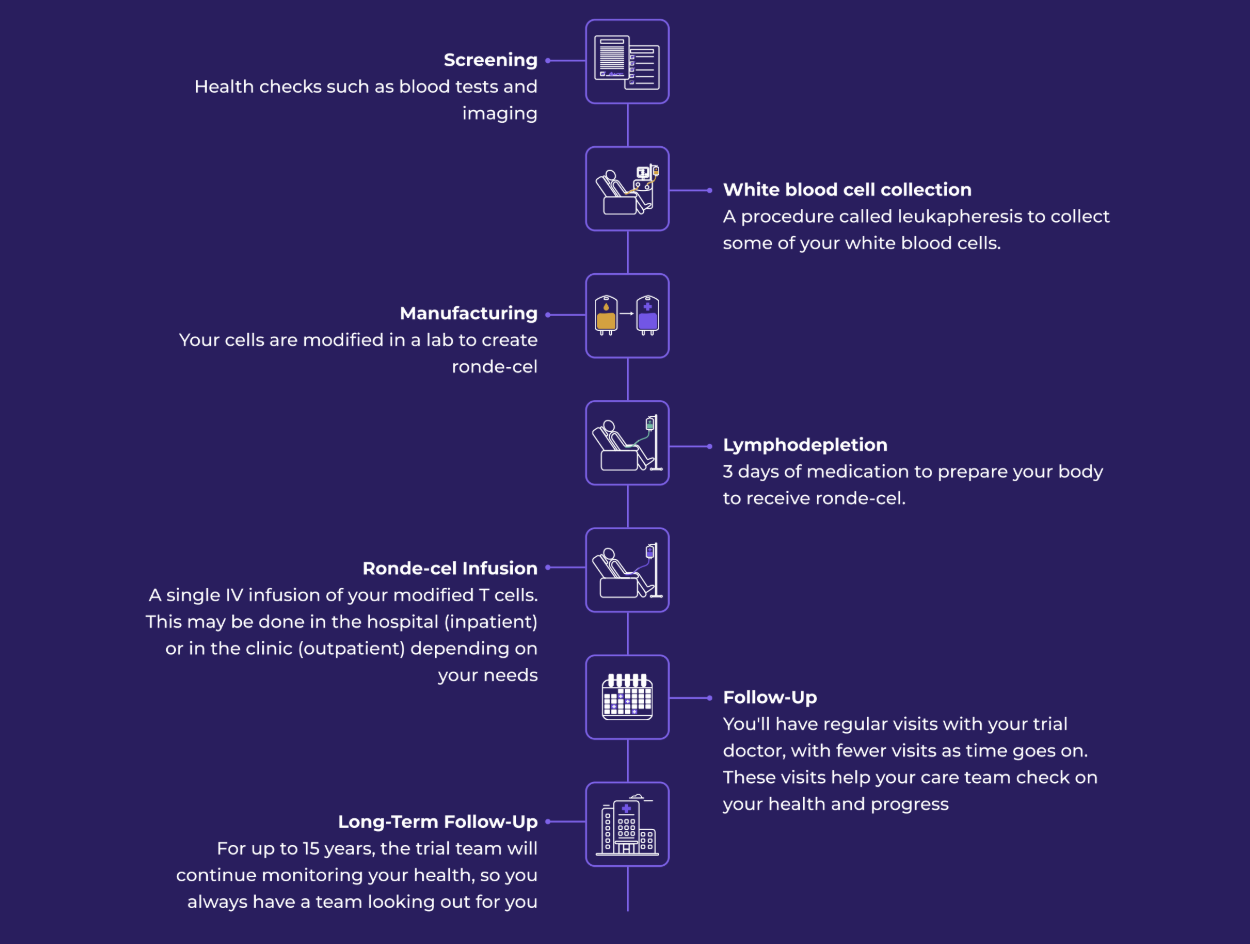
A Trial for People with Aggressive B-Cell Non-Hodgkin Lymphoma. Lymphoma is a type of blood cancer. Answer a few questions to check your eligibility.
Apply to Participate
Please fill out this form to see if you may qualify for this clinical trial and apply to participate.
Could this also be relevant to someone you know?